
Gas phase particles (atoms, molecules, or ions) move around freely in the absence of an applied electric field.
The gaseous state of matter is found between the liquid and plasma states,[2] the latter of which provides the upper temperature boundary for gases. Bounding the lower end of the temperature scale lie degenerative quantum gases[3] which are gaining increased attention these days.[4] High-density atomic gases super cooled to incredibly low temperatures are classified by their statistical behavior as either a Bose gas or a Fermi gas. For a comprehensive listing of these exotic states of matter see list of states of matter.
Physical characteristics

Drifting smoke particles provide clues to the movement of the surrounding gas.
Gas particles are widely separated from one another, and as such do not influence adjacent particles to the same degree as liquids or solids. This influence (intermolecular forces) results from the magnetic charges that these gas particles carry. Like charges repel, while oppositely charged particles attract one another. Gases made from ions carry permanent charges, as do compounds with their polar covalent bonds. These polar covalent bonds produce permanent charge concentrations within the molecule while the compound's net charge remains neutral. Transient charges exist in covalent bonds of molecules and are referred to as van der Waals forces. The interaction of these intermolecular forces varies within a substance which determines many of the physical properties unique to each gas.[5][6] A quick comparison of boiling points for compounds formed by ionic and covalent bonds leads us to this conclusion[7]. The drifting smoke particles in the image provides some insight into low pressure gas behavior.
Compared to the other states of matter, gases have an incredibly low density and viscosity. Pressure and temperature influence the particles within a certain volume. This variation in particle separation and speed is referred to as compressibility. This particle separation and size influences optical properties of gases as can be found in the following list of refractive indices. Finally, gas particles spread apart or diffuse in order to homogeneously distribute themselves throughout any container.
Macroscopic
When observing a gas, it is typical to specify a frame of reference or length scale. A larger length scale corresponds to a macroscopic or global point of view of the gas. This region (referred to as a volume) must be sufficient in size to contain a large sampling of gas particles. The resulting statistical analysis of this sample size produces the "average" behavior (i.e. velocity, temperature or pressure) of all the gas particles within the region. By way of contrast, a smaller length scale corresponds to a microscopic or particle point of view.From this global vantage point, the gas characteristics measured are either in terms of the gas particles themselves (velocity, pressure, or temperature) or their surroundings (volume). By way of example, Robert Boyle studied pneumatic chemistry for a small portion of his career. One of his experiments related the macroscopic properties of pressure and volume of a gas. His experiment used a J-tube manometer which looks like a test tube in the shape of the letter J. Boyle trapped an inert gas in the closed end of the test tube with a column of mercury, thereby locking the number of particles and temperature. He observed that when the pressure was increased on the gas, by adding more mercury to the column, the trapped gas volume decreased. Mathematicians describe this situation as an inverse relationship. Furthermore, when Boyle multiplied the pressure and volume of each observation, the product (math) was always the same, a constant. This relationship held true for every gas that Boyle observed leading to the law, (PV=k), named to honor his work in this field of study.
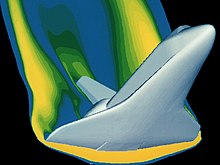
There are many math tools to choose from when analyzing gas properties. As gases are subjected to extreme conditions, the math tools become a bit more complex, from the Euler equations (inviscid flow) to the Navier-Stokes equations[8] that fully account for viscous effects. These equations are tailored to meet the unique conditions of the gas system in question. Boyle's lab equipment allowed the use of algebra to obtain his analytical results. His results were possible because he was studying gases in relatively low pressure situations where they behaved in an "ideal" manner. These ideal relationships enable safety calculations for a variety of flight conditions on the materials in use. The high technology equipment in use today was designed to help us safely explore the more exotic operating environments where the gases no longer behave in an "ideal" manner. This advanced math, to include statistics and multivariable calculus, makes possible the solution to such complex dynamic situations as space vehicle reentry. One such example might be the analysis of the image depicting space shuttle reentry to ensure the material properties under this loading condition are not exceeded. It is safe to say that in this flight regime, the gas is no longer behaving ideally.
Pressure
Main article: Pressure
The symbol used to represent pressure in equations is "p" or "P" with SI units of pascals.When describing a container of gas, the term pressure (or absolute pressure) refers to the average force the gas exerts on the surface area of the container. Within this volume, it is sometimes easier to visualize the gas particles moving in straight lines until they collide with the container (see diagram at top of the article). The force imparted by a gas particle into the container during this collision is the change in momentum of the particle. As a reminder from classical mechanics, momentum, by definition, is the product of mass and velocity.[9] Notice that during a collision only the normal component of velocity changes. A particle traveling parallel to the wall never changes its momentum. So the average force on a surface must be the average change in linear momentum from all of these gas particle collisions. To be more precise, pressure is the sum of all the normal components of force exerted by the particles impacting the walls of the container divided by the surface area of the wall. The image "Pressurized gases" depicts gas pressure and temperature spikes used in the entertainment industry.
Temperature
Main article: Thermodynamic temperature
The symbol used to represent temperature in equations is T with SI units of kelvins.The speed of a gas particle is proportional to its absolute temperature. The volume of the balloon in the video shrinks when the trapped gas particles slow down with the addition of extremely cold nitrogen. The temperature of any physical system is related to the motions of the particles (molecules and atoms) which make up the [gas] system.[10] In statistical mechanics, temperature is the measure of the average kinetic energy stored in a particle. The methods of storing this energy are dictated by the degrees of freedom of the particle itself (energy modes). Kinetic energy added (endothermic process) to gas particles by way of collisions produces linear, rotational, and vibrational motion as well. By contrast, a molecule in a solid can only increase its vibration modes with the addition of heat as the lattice crystal structure prevents both linear and rotational motions. These heated gas molecules have a greater speed range which constantly varies due to constant collisions with other particles. The speed range can be described by the Maxwell-Boltzmann distribution. Use of this distribution implies ideal gases near thermodynamic equilibrium for the system of particles being considered.
Specific volume
Main article: Specific volume
The symbol used to represent specific volume in equations is "v" with SI units of cubic meters per kilogram.See also: Gas volume
The symbol used to represent volume in equations is "V" with SI units of cubic meters.When performing a thermodynamic analysis, it is typical to speak of intensive and extensive properties. Properties which depend on the amount of gas (either by mass or volume) are called extensive properties, while properties that do not depend on the amount of gas are called intensive properties. Specific volume is an example of an intensive property because it is the ratio of volume occupied by a unit of mass of a gas that is identical throughout a system at equilibrium.[11] 1000 atoms of protactinium as a gas occupy the same space as any other 1000 atoms for any given temperature and pressure. This concept is easier to visualize for solids such as iron which are incompressible compared to gases. When the seat ejection is initiated in the rocket sled image the specific volume increases with the expanding gases, while mass is conserved. Since a gas fills any container in which it is placed, volume is an extensive property.




No comments:
Post a Comment